Scientists Discover Way To Leverage High-Energy Sunlight for Fuel Production
Semiconductor-Catalyst Combo Captures Energy To Drive Chemical Reactions
Plants and algae make their fuel from sunlight. Perhaps we could do the same using semiconductors. A team of scientists at the National Laboratory of the Rockies (NLR) made strides in that direction.
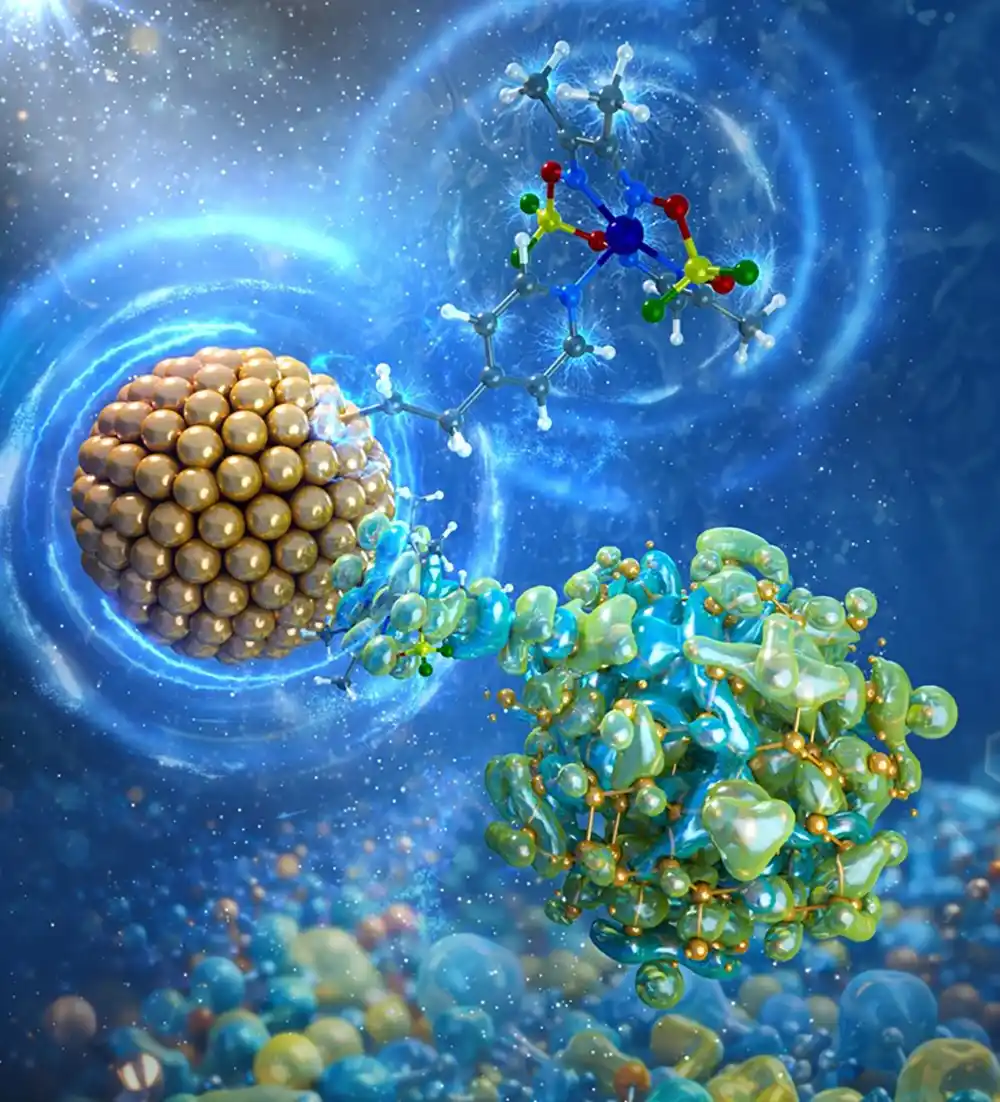
They discovered a silicon semiconductor coupled to a molecular catalyst can capture higher-energy sunlight that is unused by both plants and human-made panels. Such energy could be used to drive reactions, like that between carbon dioxide and water to form hydrocarbon fuels and chemicals, or that synthesize fertilizer from nitrogen gas, which makes up 20% of our atmosphere.
This work—which touches the fields of artificial photosynthesis and photocatalysis—was recently published in the Journal of the American Chemical Society in an article titled, “High-Energy Hybridized States Enable Long-Lived Hot Electrons in Cobaloxime-Silicon Nanocrystal System.”
“Our work seeks to push the limits of how much energy we can yield from the sun, and the semiconductor-molecular catalyst hybrid system used in this study reveals one possible pathway,” said Nathan Neale, research scientist at NLR and the paper’s lead author. “We found electronic states in this hybrid system keep photogenerated electrons energetic long enough for use in chemical reactions.”
A motivation of this work is that sunlight has more energy available than we currently use. For example, solar panels might use around 20% of the energy in the incident light. Plants and other photosynthetic organisms might use just 1%. In both cases, sunlight transfers its energy to electrons, with the higher-energy electrons quickly losing much of their absorbed energy in the form of heat, which results in low efficiency.
“High-energy electrons often lose their energy very rapidly in materials by coupling with molecular vibrations and heating up their surroundings,” Neale said. “By blending electronic states between the light-harvesting silicon semiconductor and the molecular catalyst, our material kept the electrons ‘hot’ for at least five nanoseconds, which potentially could be used to drive photocatalysis at superior efficiency.”
Although nanoseconds are brief, they are much longer than the tens of femtoseconds typically observed for electron cooling. In fact, the high-energy electrons in this study stayed “hot” for approximately 25,000 times longer than the typical amount of time it takes hot electrons to cool down in silicon.
Keepin’ It Hot
The researchers achieved these longer electron lifetimes by manipulating the molecular chemistry at the semiconductor surface. The important factor is the linking group, an ethylenepyridine unit. This unit fused the silicon nanocrystal to the catalyst and enabled the formation of a hybrid electronic state that allowed the electrons to persist. This revelation about the role of the ethylenepyridine linker compound is a new way of thinking about these molecular bridges.
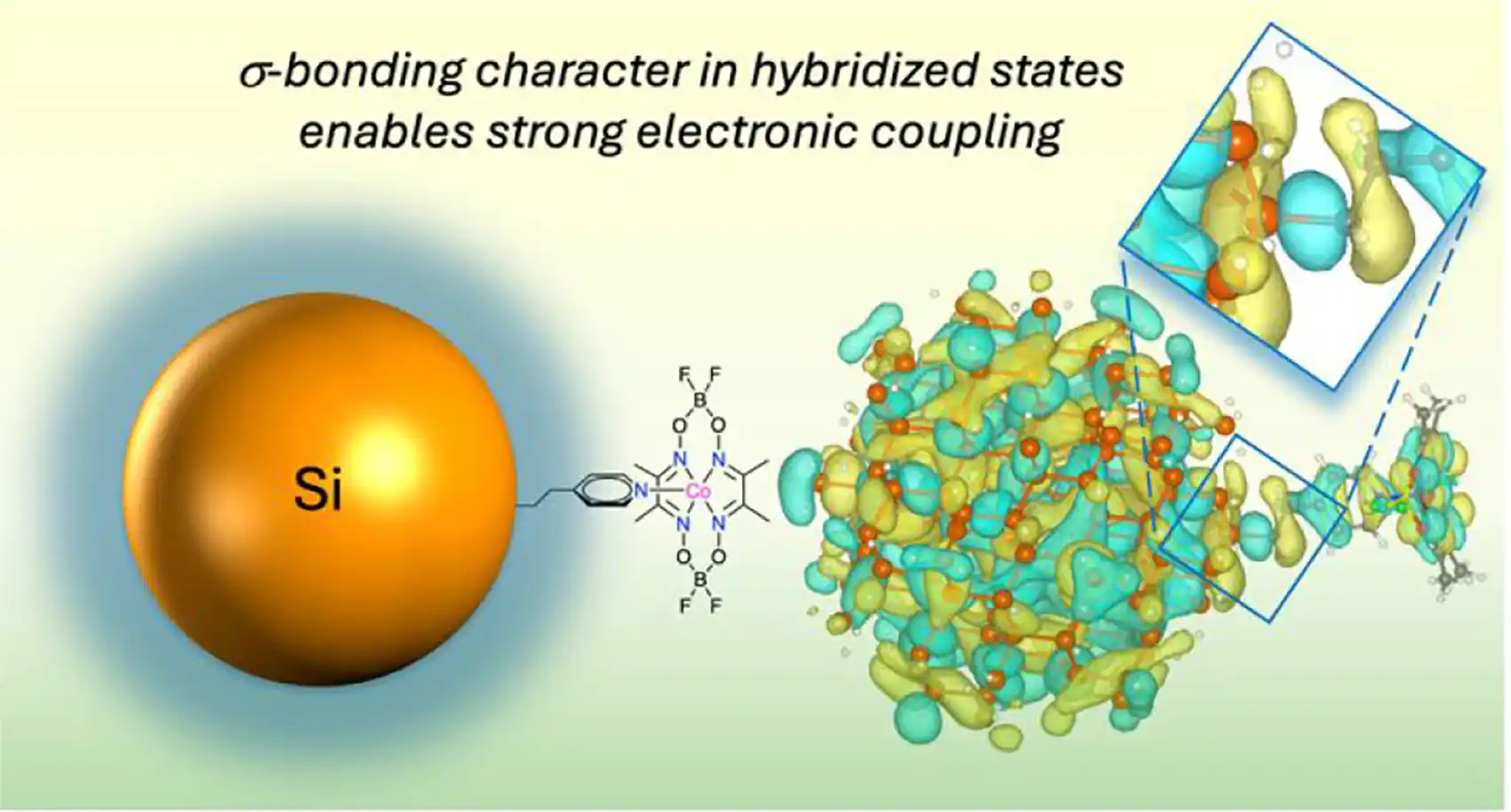
“The extreme sensitivity to the linking group chemistry teaches us that it is insufficient to simply provide a spatial proximity between a semiconductor and a surface-bound catalyst to achieve efficient photoinduced processes,” the researchers stated in the conclusion of the study.
Neale’s team confirmed the role of the molecular tether by using several spectroscopy methods to study the semiconductor/catalyst hybrid. Next, they performed quantum mechanical calculations to model the exact photoelectronics. They discovered that the blended electronic states allow the hot electrons to spread out in both the silicon and catalyst.
Fuels, Fertilizers, and Beyond
Direct sun-to-fuel semiconductors are not mainstream energy products. But this work builds on widespread research to demonstrate that such new technology is feasible. By using these findings to keep electrons hot longer, engineers could split water to create hydrogen, or carbon dioxide to create hydrocarbon fuels, and harvest more energy.
This work was supported by the U.S. Department of Energy’s Office of Science Basic Energy Sciences program and performed in labs at NLR.
Learn more about NLR’s work in photochemistry and basic energy sciences and how to partner with the laboratory on chemistry and nanoscience research. Read “High-Energy Hybridized States Enable Long-Lived Hot Electrons in Cobaloxime-Silicon Nanocrystal System” in the Journal of the American Chemical Society.
Last Updated April 28, 2026
